

P = Pressure (atm) V = Volume (L) n = moles R = gas constant = 0.0821 atm R is the universal gas constant measured in J/(K. The units used in the ideal gas equation that PV = nRT are: Atmosphere in which we live has normally 1 atm pressure. In chemistry, ATM is a unit to measure pressure. … Thus, in the given reaction, 340 Atm means that the said amount of pressure is required for converting the reactants into the products.
#Convert 745 mmhg to atm full
What is the full form of 340 atm?Ītm stands for atmospheres, which is a unit for measuring pressuring. … It is approximately equal to Earth’s atmospheric pressure at sea level. The standard atmosphere (symbol: atm) is a unit of pressure defined as 101325 Pa (1.01325 bar).

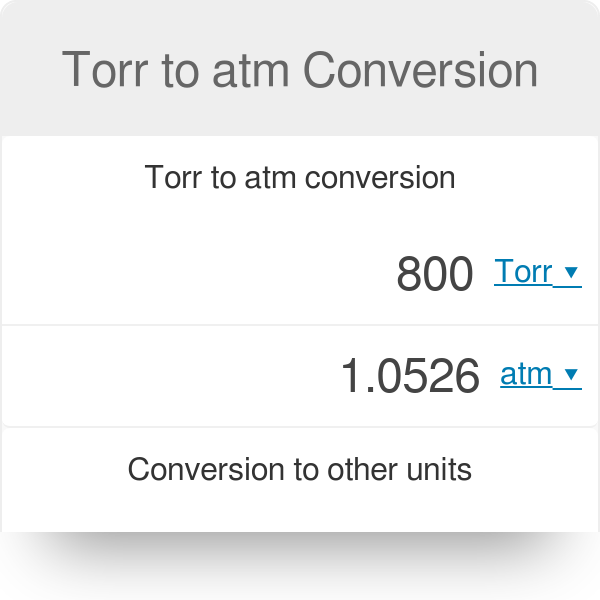
This means that to convert mmHg to atm you should multiply your figure by 0.0013157896611399. One millimeter of mercury is equal to 0.0013157896611399 atmospheres. How do you convert mmHg to Torr?ġ mmHg = 1.00 torr.
The value of R depends on the units involved, but is usually stated with S.I. The ideal gas law is: pV = nRT, where n is the number of moles, and R is universal gas constant. Liter atmosphere is a unit of energy, symbole “l-atm”, equal to 101.32500 joules. T = Temperature expressed in units of Kelvin. PV = nRT is an equation used in chemistry called the ideal gas law equation. It is defined as the pressure exerted by the weight of the atmosphere, which at sea level has a mean value of 101,325 pascals (roughly 14.6959 pounds per square inch). How many torr are there if there is 3.5 atm?Ītm in chemistry stands of atmospheric pressure. Which value of R will you use if the pressure is given in ATM?.How do you convert ATM liters to calories?.What are the advantages to the customer of using an ATM?.How many torr are there if there is 3.5 atm?.


 0 kommentar(er)
0 kommentar(er)
